Alright, get all your fertilizers together and we are going to design a fertilizer program for cannabis. Many of you know how to plug in figures on the computer but I just want to spend some time to design the program.
You may use the table of different fertilizers I included in my last article or use the labels on the fertilizer bags you’re using. Also note that numbers on the fertilizer bags show per cent nitrogen, per cent phosphate (P2O5) and potash (K20). Our calculations are in per cent nitrogen, per cent phosphorus and per cent potassium. I am using 100 litres of water for calculation
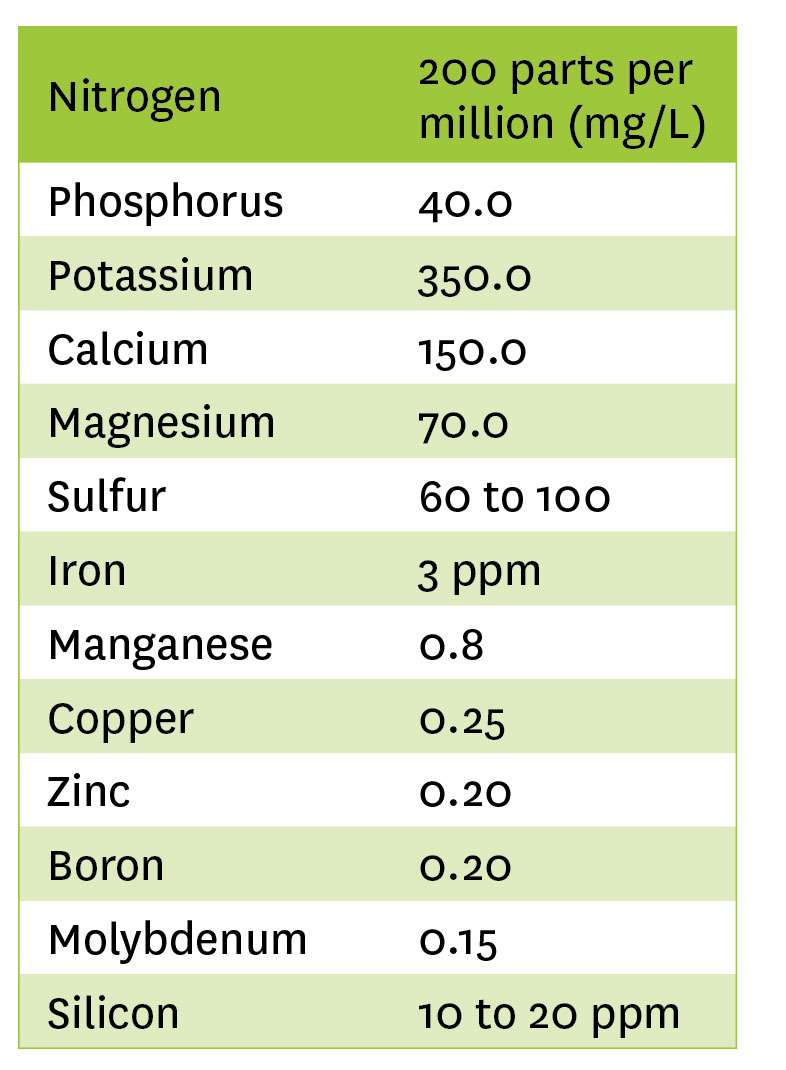
This is a typical full feed for cannabis. Adjustments are made based on plant growth and any deficiencies which may occur due to pH of the growing medium. Also, in this formulation, there is only one per cent ammonium nitrogen coming from calcium nitrate; the rest is all nitrate nitrogen.
Step 1: Remember the formula
- ppm required x water volume in Litres ÷ % purity of fertilizer x 10 = grams of fertilizer
- grams of fertilizer x % purity of fertilizer x 10 ÷ volume of water in L = ppm
Step 2: Start with taking all the calcium from calcium nitrate, 15.5-0-0 + 19.8% calcium. Using the above formula:
- 150ppm of calcium needed x 100L of water ÷ 19.8% calcium x 10 = 75.75 grams
- Since it is 15.5% Nitrogen, how much nitrogen do we have in 75.75 grams? Using Step 1, b: 75.75 x 15.5 x 10 ÷ 100L = 117.4ppm of N
- We need 200 N, so 200 – 117 = 83ppm N needed
Step 3: Take all your nitrogen from Potassium nitrate 13-0-46 (13-0-38, NPK)
- 83 x 100L ÷ 13 x 10 = 63.8 grams. How much potassium do I have?
- 63.8 x 38 x10 ÷ 100 = 242.4ppm of K
- Balance of K needed is 350, so 350 – 242.4 = 107.6ppm K needed
Step 4: Now take all your phosphorus from mono potassium phosphate (0-53-34), which is 0-22.79-28.22 as NPK. We need 40 ppm of P
- 40 x 100L ÷ 22.79 x 10 = 17.6 gram. How much K did we get from this?
- 17.6 x 28.22 x 10 ÷ 100L = 49.28ppm K.
- Total K (above) is 242.4, so: 242.4 + 49.28 = 291.68. We need 350ppm, so: 350 – 291.68 = 58.32ppm K needed
Step 5: Take 58.32ppm from potassium sulfate, 0-0-52 or 0-0-41.5% K and 18% S. You can do it now. If not, then use the same formula:
- 58.32 x 100L ÷ 41.5 x 10 = 14.0 grams. How much S have we got?
- 14.0 x 18.0 x 10 ÷ 100 = 25.2 ppm S
Step 6: Now take 70 ppm of magnesium from Magnesium sulfate, 0-0-0-10% Mg and 12% sulfur. Some growers prefer to use magnesium nitrate.
- 70 x 100 ÷ 10 x 10 = 70 grams. How much sulphur have we got from this?
- 70 x 12 x 10 ÷ 100 = 84 ppm S
- Total sulfur is 84 + 25.2 = 109.2
- You may have noticed that sulphur levels could vary based on the use of potassium sulfate and magnesium sulfate. You can bring it down by using magnesium nitrate but then nitrogen will have to be adjusted.
Now we are ready to calculate trace or micro elements. They are single elements so it’s a little easier to calculate.
Step 7: Take 3ppm of iron from 13% iron chelate. If you are using iron sulfate then know the concentration of iron and sulfur.
- 3ppm needed x 100L of water ÷ 13% x 10 = 2.30 grams
Step 8: Take 0.8 ppm of manganese, 13% manganese chelate. Manganese sulfate is also available.
- 0.8 x 100 ÷ 13 x 10 = 0.61 grams
Step 9: Take 0.25 ppm of copper from 14% copper chelate. Copper sulfate is also available.
- 0.25 x 100 ÷ 14 x 10 = 0.18 grams
Step 10: Take 0.20 zinc from 14% zinc chelate. Zinc sulfate is also available.
- 0.20 x 100 ÷ 14 x 10 = 0.14 grams
Step 11: Take 0.2 ppm from Borax, 15% B
- 0.2 x 100 ÷ 15 x 10 = 0.133 grams
Step 12: Take 0.15 ppm of molybdenum from sodium molybdate 46%. Many other formulations are available.
- 0.15 x 100 ÷ 46 x 10 = 0.032 grams
Step 13: My recommendation is to use silicon. It provides many benefits to cannabis plant health including reduced levels of powdery mildew. Depending on the source you want to use, make your calculations.
There are many aspects of fertilizer use: what to mix or not to mix, making stock solutions of trace elements and general cannabis plant management. I will address those aspects in a future article.
Moyhuddin Mirza, PhD, is chief scientist with the Cannabis Nature Company in Edmonton, and a consultant with the cannabis industry. Email him at drmirza@cannabisnature.ca
Print this page