
The cannabis plants’ needs are pretty basic, compared to other crops — like poinsettia, for example, which needs higher levels of molybdenum, or peppers with their higher boron requirement.
In my research with cannabis and hemp, I have not found any special requirement of an element needed for optimum growth of cannabis. However, in our research comparing different nitrogen to potassium ratios, we found that increasing the ratio to 1:2 at bud stage significantly increased CBD content.
Many ready-made fertilizers available in the market offer different types of claims and are generally very expensive. Growers should do their due diligence on these claims and use science rather than emotions to decide cost and benefit ratios.
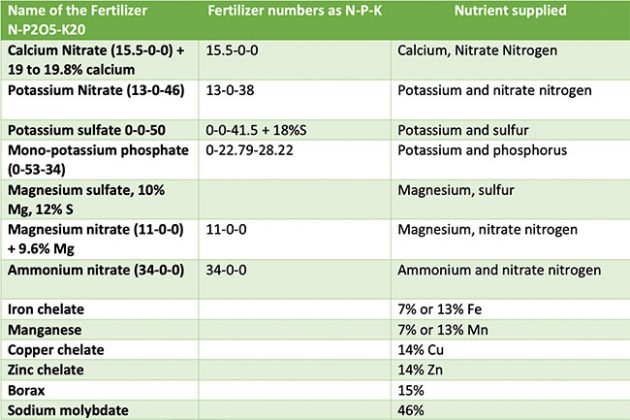
The chart outlines the basic ingredients for designing your cannabis fertilizer and nutrient program.
The numbers on the fertilizer bags indicate percentages of nitrogen, phosphate P2O5 and potash K20. Actual calculations are made based on phosphorus and potassium. In order to obtain the percentage of phosphorus, multiply the middle number with 0.43 and last number by 0.83.
The numbers on the label can vary slightly based on the fertilizer manufacturer. For example, potassium nitrate could also be 13-0-44 rather than 13-0-46
Fertilizers used for cannabis must be highly soluble and greenhouse grade.
When preparing stock solutions, calcium nitrate is kept separate from all other fertilizers because calcium should not be mixed with fertilizers containing phosphates and sulfates in stock solutions.
Know your injectors, how do they work and how different dilution ratios can be changed to get the desired electrical conductivity (EC). I recommend having at least four injectors. One for calcium nitrate, the second for all the other macros – potassium, phosphorus, magnesium and ammonium. The third injector is for all the trace elements: iron, manganese, copper, zinc, boron and molybdenum. This gives you the benefit of not reducing the concentration of these trace elements when changing the EC. The fourth injector is for pH control.
Be aware that chelated nutrients are more stable for uptake by cannabis plants at different pH ranges. All trace elements are also available in other forms; for example, iron sulfate, manganese sulfate, copper and zinc sulfates.
It is important to know how to calculate parts per million (ppm). PPM is a measure of concentration and is a common unit used in cannabis fertilizer management, although some growers use micromoles and millimoles as well.
Basically, one part of a chemical in a million part of water or other solvent is a ppm. Many growers are afraid of math in making ppm calculations because of fear of miscalculations. But fear not because plants will forgive you, within certain limits, and plants have their own mechanisms to absorb nutrients when they need it and how much. Our job is to supply those nutrients with every irrigation in the root zone.
Some good formulas for calculating ppm are available on the internet. The formula I have used for 40 years is:
ppm x water in L ÷ % purity of fertilizer x 10 = grams of fertilizer
or
grams of fertilizer x % purity of fertilizer x 10 ÷ water in L = ppm
For example: You have calcium nitrate fertilizer, 15.5-0-0 + 19.8% ca. You need 150 ppm of calcium in 100 L of water. So 150 x 100 ÷ 19.8 x 10 = 75.75 grams.
Then we need to know how much nitrogen we got from calcium nitrate, so 75.75 x 15.5 x 10÷100 L = 117 ppm of N.
For a simple rule-of-thumb to calculate ppm fast, simply remember: one gram/L. So, if you dissolve one gram of a chemical/fertilizer in one L of water, then just multiply the % of that chemical with 10 and you will get ppm.
If you take one gram of calcium nitrate 15.5-0-0 + 19.8% ca, then at one gram/L of water, you will get 155 ppm of nitrogen and 198 ppm of calcium. That should be easy to remember.
Moyhuddin Mirza, PhD, is chief scientist with the Cannabis Nature Company in Edmonton, and a consultant with the cannabis industry. Email him at drmirza@cannabisnature.ca
Print this page
